The northern hemisphere faces a bleak winter of rising Covid-19 cases, but signals that two rapidly engineered vaccines by Pfizer and Moderna work on 95 per cent of candidates on whom they have been tested have prompted joy.
The results are interim ones but they are, nevertheless, figures from large-scale Phase 3 clinical trials. The first 40 million doses could be administered in the United States by the year end.
The other leading contender, Oxford University-AstraZeneca boosted the public mood also when it released less definitive, but still encouraging, Phase 2 findings on its vaccine. Causing few side effects, particularly among older people, it prompts strong responses in innate immunity – the immunity with which a body is born and adaptive immunity, that acquired after exposure to disease – even with low doses.
Even the makers of the Sputnik V vaccine, much promoted by Moscow earlier this year, has now sought certification from the World Health Organisation, suggesting they are confident it can stand up to international challenge.
It is hard not to feel a sense of optimism. Moderna’s vaccine has a confirmed 94.5 per cent efficacy; Pfizer-BioNTech hits 95 per cent on that scale, while it also works safely on the over-65s.
The success rates are significant. Remember, it is a good year for a flu vaccine when it works on 55 per cent of those who go for jabs but even that outcome is enough to prevent a blight becoming a plague.
Whether a vaccine works is one thing, whether it lasts is another. Soon, we may know the answer to that also. Early indications from the US La Jolla Institute of Immunology suggest it may be years, even decades.
Final results may not be as good as first thought but the bottom line for those eager for a vaccine, and those doubtful, is that we will soon know either way. Clear results from at least four lead candidates will be known by Christmas.
Vaccine tracker
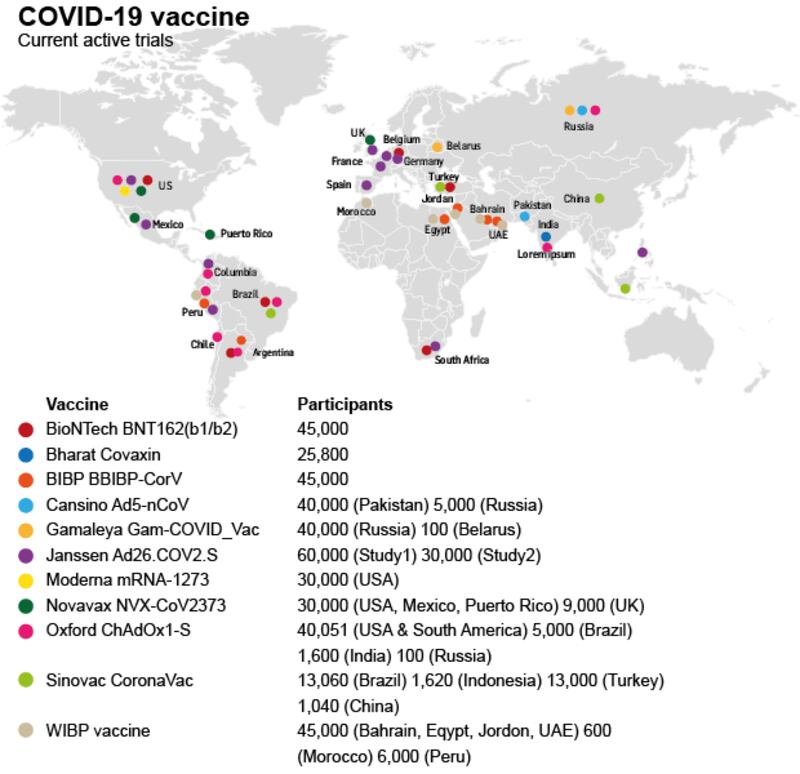
Twelve vaccines are in Phase 3 trials, each conducted on up to 60,000 people. Six from China and Russia are approved for early/limited use but without final Phase 3 results.
Fifty-four more are in smaller, earlier Phase 1 and Phase 2 trials. Eighty-seven others are in so-called preclinical trials, being tested on animals but not yet on humans.
Vaccines can be authorised during emergencies ahead of formal approval. To speed things up leading contenders are already being made, but safety research continues afterwards.
Contenders: On what we know, the leading candidates are:
- Moderna (US): Limited quantities in the US by end of 2020, globally from next April;
- Pfizer-BioNTech (US-Germany): Probably available in limited quantities by end of the year; requested emergency authorisation from the US Food and Drug Administration on Friday;
- Oxford University-AstraZeneca (UK): Working alongside the Jenner Institute – headed by Irish scientist Prof Adrian Hill. Phase 3 results due within weeks. All going well, four million doses could be ready by the end of the year;
- Novavax (US): Phase 3 trials are progressing. Supplies predicted from the middle of next year;
- Janssen-Johnson & Johnson (US): Its second phase 3 trial began this week - it could be available by the second half of 2021;
- Valneva (France): It is hoping for regulatory approval by the second half of 2021;
- Sinovac (China): Mid-stage trials have been successful. On the back of Phase 3 trials under way, news is due shortly on when it could be ready;
- Sanofi-GSK (France): Phase 3 trials start next month with 30,000 volunteers in different countries. Approval expected by June 2021.
The technology
Among leading vaccine candidates, four different types emerge:
mRNA: This uses genetic material known as mRNA; the genetic code inside the coronavirus responsible for its spike protein is identified.

The vaccine injects copies of the spike code into human cells which then produce spike protein. It covers the surface of the virus, training the body to produce antibodies and other defences – deployed by Pfizer-BioNTech and Moderna.
Adenovirus: The genetic code that produces the spike protein of the coronavirus is identified and added to a different benign common-cold virus, which prompts cells to make antibodies – deployed by Oxford-AstraZeneca and Janssen.
Viral protein: A virus is used to insert the genetic code for the spike protein into the cells of an insect, which produces the spike protein. This is harvested, an "adjuvant" is added and the combination stimulates the immune system – deployed by Novavax and GSK-Sanofi.
Inactive virus: Coronavirus particles are inactivated using a chemical that keeps spike protein intact. The "dead" virus is injected into the body, prompting an immune response – deployed by Valneva
This is not a one-horse race and none are fully approved, says Dr Siobhán McClean of UCD School of Biochemistry, who notes that mRNA vaccines are unstable and must be chilled but are easier and quicker to make.
Unlike Pfizer-BioNTech’s offering, Moderna’s does not have to be stored at -70 degrees. It tolerates -20 degrees; the standard for hospital and pharmacy freezers. So it should be easier to distribute and store.
The -70 degree vaccines, nonetheless, are suitable for hospital workers for example. By contrast, the Oxford vaccine may be more stable: “Being more robust, it has a lot to offer,” she says.
Vaccine specialist Dr Anne Moore of UCC, who previously worked with the Oxford team, says there are no huge problems with adenovirus-based vaccines but some concern about their efficacy if distributed globally.
The key question is how little, or how much is needed to provoke immunity. That will show how long vaccines will last and whether they are safe for the elderly. “We should have some good vaccines,” she predicts.
Timelines
The Health Service Executive faces a huge logistical task when the vaccines are ready since double doses will be needed in most cases. Fourteen billion doses are needed globally.
If vaccines stop 90 per cent of infection, herd immunity is realisable. For Sars-CoV-2 the herd immunity threshold is estimated to be about 70 per cent. So an 80 per cent vaccination rate could be enough.
In an age of anti-vaxxers, however, public trust will have to be won. Some may run into difficulties with a few or more patients when they are given to millions, not just tens of thousands in tests.
Either way, they will not be a silver bullet, according to the British Academy and the Royal Society which says safety fears must be addressed and disinformation challenged. "Life will not immediately get back to normal," it says.
With enough supplies, healthcare workers and the vulnerable in Ireland could be covered by the summer and everyone by next September. But that will require "massive immunisation clinics", cautions Dr McClean.
By the time supplies are ready “there will be a lot more data available to allow people make the most informed decision that they can. So don’t make up your mind yet,” Moore advises doubters.
All too rushed?
For the uneasy, the developer of the Oxford vaccine, Prof Sara Gilbert says progress has been fast because "so much work on the technology" required was done before work on the virus began.
“So people shouldn’t be worried we’re going too quickly. We’re being very careful to assess safety at each stage . . . This is always our top priority and we’ll continue to do that,” she says.
Available doses
Ireland is part of the European Commission’s vaccines order, which spreads the risk faced by individual EU states by linking countries to multiple vaccines, not just one and by offering purchasing clout.
Deals are signed with Oxford-AstraZeneca, Sanofi-GSK and Janssen, while “finalised contract negotiations” are under way with Pfizer-BioNTech and Moderna to provide 460 million doses next year. Ireland will get 4.6 million doses.
"The virus will not be disrupting us for years," says Dr Moncef Slaoui, chief scientific adviser for the US public-private partnership Operation Warp Speed which is tasked with developing and distributing Covid vaccines.
“If enough of us get vaccinated, there is a good chance that we keep this virus at very, very low levels, which means we can go back to normal life, fully normal life,” he says.
The big question remains about which of the vaccines will work best. So the more we have and know, the better.










