Early in 2020, Liz McMahon and David Ryan received news that would dramatically change the life of their youngest son.
“You have won the lottery,” Dr Declan O’Rourke, a consultant paediatric neurologist at Temple Street children’s hospital, told them.
Seán Ryan, then aged seven weeks, had been selected for participation in a rapid access programme for a new one-off gene replacement therapy that would treat his Spinal Muscular Atrophy (SMA) before symptoms had begun to show. In the most severe cases, the rare and progressive muscle-weakening caused by SMA stops children from walking and creates problems for breathing and swallowing. Many of these children do not live past the age of two.
“SMA is a degenerative disease, so the earlier we give this treatment the better the clinical outcome. Children we can treat pre-symptomatically ultimately do best,” explains Dr O’Rourke.
Seán was among the first four children in Ireland to undergo gene replacement therapy as part of a Zolgensma trial at St James’s hospital in Dublin. Zolgensma, one of the most expensive drugs in the world, costs more than $2 million (€1.7m) for one-off treatment in the United States. Last week, the Health Service Executive announced it was approving it for use under reimbursement for very young patients with severe SMA.
The trial involved a ground-breaking collaboration between St James’s and Temple Street hospitals and researchers at Trinity College Dublin led by Prof Martina Hennessy. She had the previous year conducted the first gene replacement clinical trial at St James’s on an adult patient with haemophilia. However, the Zolgensma trial would be the first time babies were treated at the site, taking place while the country’s hospitals were gripped by the Covid-19 pandemic.
McMahon recalls how Seán’s treatment was almost thwarted by the pandemic. The drug was flown to Ireland on one of the last flights from the US before then president Donald Trump imposed a travel ban.
Seán’s treatment was brought forward to March 16th, 2020, “[as if it was] meant to be”, says McMahon. “It was nail-biting, panic station stuff. Going to St James’s was just an amazing feeling. To be there knowing what everyone had done to make this possible for Seán.”
Seán was diagnosed with SMA prior to his birth. At 10 days old he began treatment with Spinraza, another new drug injected into the spine at least every four months to prevent deterioration. Now with Zolgensma, McMahon says she and her husband often forget that their one-year-old has what would have been a life-limiting condition.
The youngest of three children, Seán owes his early diagnosis to the difficult journey charted by his older brother, Luke (3).
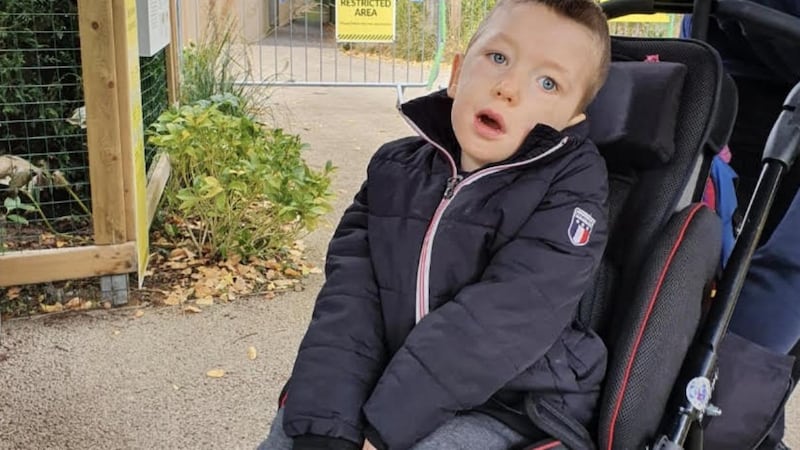
Speaking from Temple Street, where Luke has been admitted with the flu, McMahon says SMA is all-consuming in his life. It took weeks (a long period in the SMA lifecycle) for Luke to receive a diagnosis. By that stage he had lost muscle tone and couldn’t lift his arms.
At six weeks Luke caught bronchiolitis which led to severe respiratory distress. He was put on a ventilator before being intubated and he spent eight months in hospital.
“It is very distressing to see your tiny little baby with doctors all over him. It was very tough,” his mother recalls.
When not in hospital, Luke requires a night nurse seven nights a week. He uses a ventilator to sleep and needs a nebuliser and chest physiotherapy each morning. Although his intellectual capacity is unaffected, muscle weakening in his face means he is largely non-verbal and finds it difficult to communicate when he is in pain. He cannot stand or walk and muscle laxity in his back has led to scoliosis and the insertion of metal rods to help straighten his spine.
Luke is too old for Zolgensma, which is currently only cleared for use in children under the weight of a typical two-year-old. Current treatments cannot undo harms already caused by SMA, but his condition has been prevented, for the time being, from severe worsening by Spinraza injections.
“Spinraza has given him a much better quality of life, but he is still very compensated,” says McMahon, adding that the spinal injections every four months cause him huge distress.
The two brothers had been attending medical assessments together, but it was “not fair” for Luke to hear the incredible progress being made by his younger sibling, says their mother.
“When you are looking at Seán and then at Luke, it is like day and night. We are always celebrating how well Seán is doing, but you nearly feel guilty at the same time.
“Luke will never walk, and I know he can see Seán walking around, and that can be very tough,” she says.
New drugs developed in the past few years have “completely changed the landscape” for children with the condition, says Dr O’Rourke.
“I have been looking after children with spinal muscular atrophy for many years. For a very long time we had no treatments to offer children beyond supportive management,” he explains.
Now doctors can offer three therapies, the most notable of which involves gene replacement therapy, which was “the stuff of science fiction” when he was in medical school, he says.
McMahon hopes the HSE will now introduce a check for SMA as part of the postnatal heel-prick test to identify health conditions that must be picked up early.
Her younger son had the benefit of early diagnosis, but if SMA was included in the postnatal check, Luke too could have started Spinraza prior to showing symptoms and “wouldn’t have had to go through all this pain”.










